Spike protein igg antibody test12/20/2023  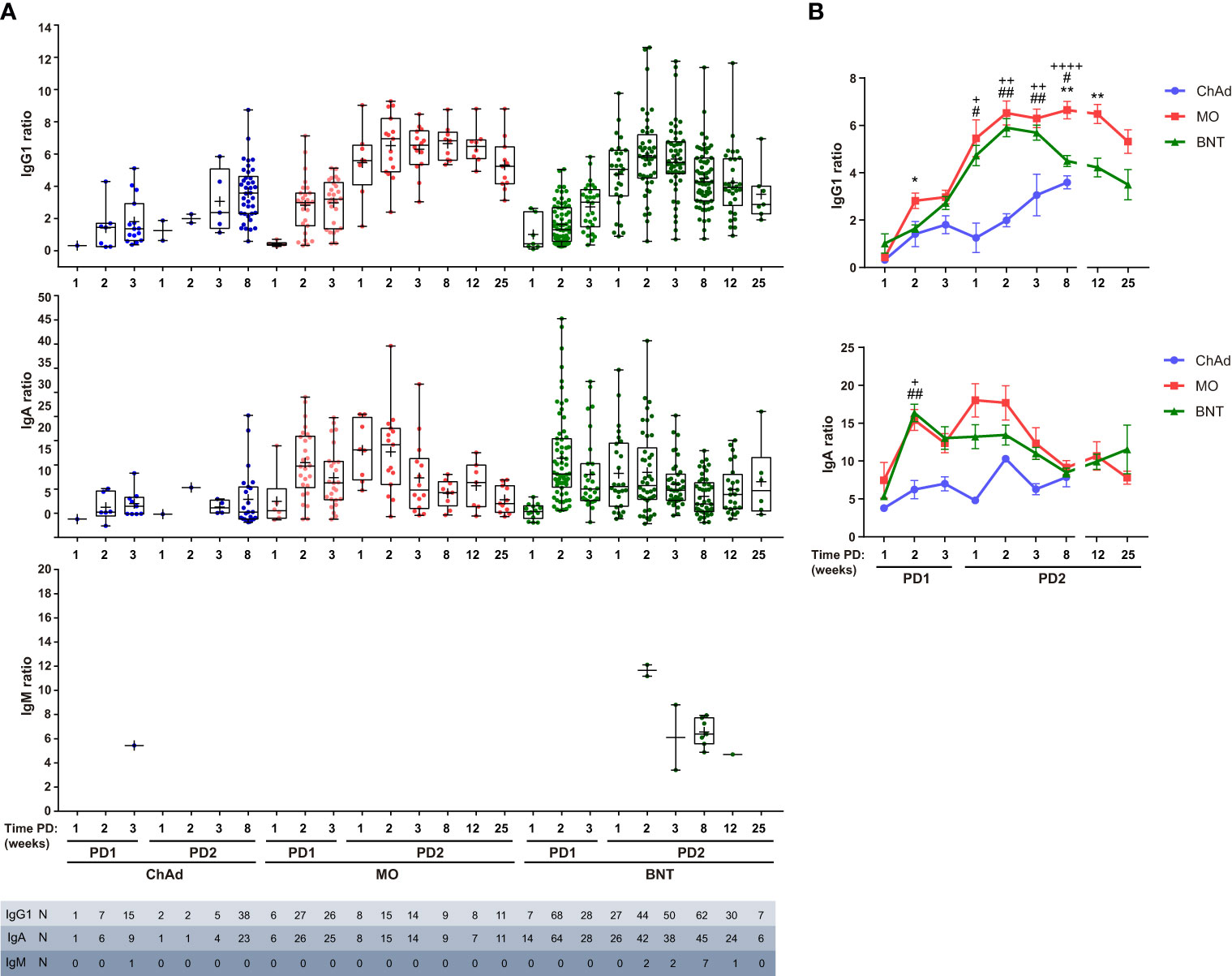
Among them, enzyme linked immunosorbent assays (ELISA), chemiluminescent immunoassays, and neutralization assays( Muruato et al., 2020) are reliable but necessitate of trained operators and require hours or even days to perform the analysis( John Hopkins Center for Health Security, 2020). Several serological tests have received the Emergency Use Authorization (EUA) from the U.S. Finally, the antibody tests can help to track how effectively the patient’s immune system is fighting the infection and are potentially helpful for plasma transfusion therapies( Krammer and Simon, 2020, Winter and Hegde, 2020, Long et al., 2020, Roback and Guarner, 2020, Duan et al., 2020, Amanat et al., 2020, LaMarca et al., 2020, Lee et al., 2020). Even though it takes several days to develop sufficient amount of antibody in blood plasma or serum once a patient is infected by SARS-CoV-2, serological analysis is crucial for the identification of asymptomatic infections to further control the spread of the virus( Paiva et al., 2020, Du et al., 2020, Cui and Zhou, 2020, Day, 2020). In addition, sampling for antibodies are easier because antibodies are more stable than RNAs. While quantitative reverse transcription polymerase chain reaction (qRT-PCR) is the most reliable method to detect the genome of SARS-CoV-2 at the early stage of the infection( Corman et al., 2020, Chu et al., 2020, Moitra et al., 2020), serological tests for viral antibodies are equally important as they can identify false negative qRT-PCR responses because the virus concentration tends to become low at the late stage of the infection( LaMarca et al., 2020). This strongly motivates the researchers to develop reliable testing tools to make SARS-CoV-2 diagnostics easier, cheaper and more accessible( Dincer et al., 2019, Choi, 2020, Morales-Narváez and Dincer, 2020, Zhu et al., 2020, Santiago, 2020, Seo et al., 2020, Bhalla et al., 2020).  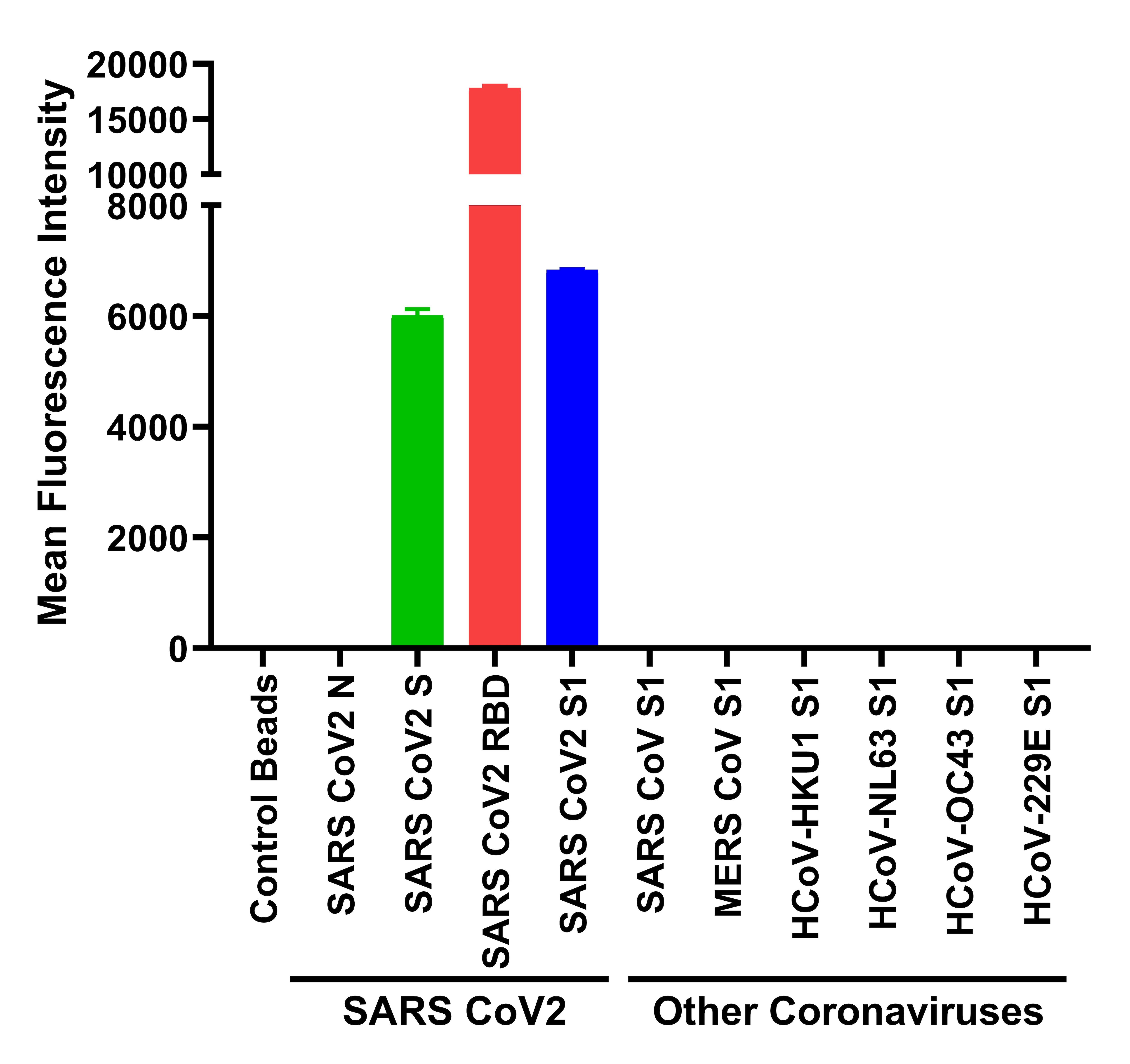
The World Health Organization (WHO) advised the international community to perform extensive diagnostic tests to reduce the spreading of the virus and decrease the number of unreported cases (i.e.,asymptomatic or mild cases)( Li et al., 2020, Zhao et al., 2020). The recent worldwide outbreak of severe acute respiratory syndrome related coronavirus 2 (SARS-CoV-2)( Wu et al., 2020) has led to unprecedented pressure on national healthcare systems( Fauci et al., 2020). We demonstrate that our opto-microfluidic platform offers a promising point-of-care testing tool to complement standard serological assays and make SARS-CoV-2 quantitative diagnostics easier, cheaper, and faster. This label-free microfluidic platform achieves a limit of detection of ∼0.08ng/mL ( ∼0.5pM), falling under the clinical relevant concentration range. The target antibody concentration can be correlated with the LSPR wavelength peak shift of gold nanospikes caused by the local refractive index change due to the antigen–antibody binding. In this work, based on the principle of localized surface plasmon resonance (LSPR), we develop an opto-microfluidic sensing platform with gold nanospikes, fabricated by electrodeposition, to detect the presence and amount of antibodies specific to the SARS-CoV-2 spike protein in 1 μL of human plasma diluted in 1mL of buffer solution, within ∼30min. While quantitative reverse transcription polymerase chain reaction (qRT-PCR) is the most reliable method to detect viral genes of SARS-CoV-2, serological tests for specific antiviral antibodies are also important as they identify false negative qRT-PCR responses, track how effectively the patient’s immune system is fighting the infection, and are potentially helpful for plasma transfusion therapies. The ongoing global pandemic of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has led to active research in its associated diagnostics and medical treatments. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
